Pfizer Release Adverse Events Data – Insane!
It was the FDA’s proposal to make available to the FOIA requester 500 pages a months. 360,000 pages… Do the math.
The FDA’s excruciatingly slow release of data related to Pfizer’s COVID-19 vaccine has already borne fruit, and it’s damning despite a trickle of just 500 pages per month out of 329,000 pages – which will take until 2076 to complete.
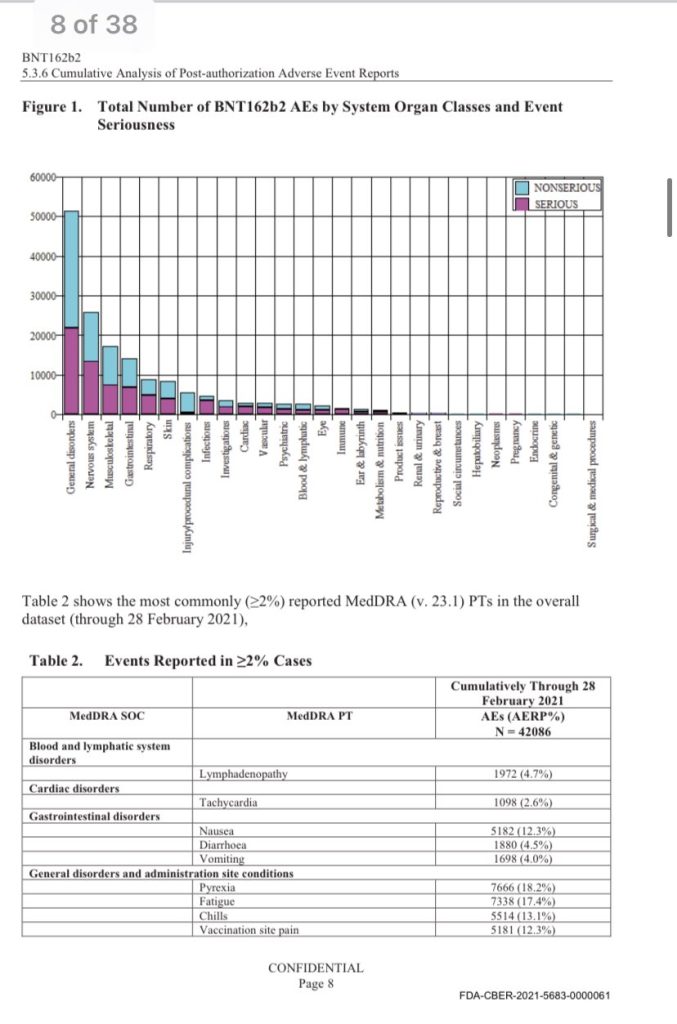
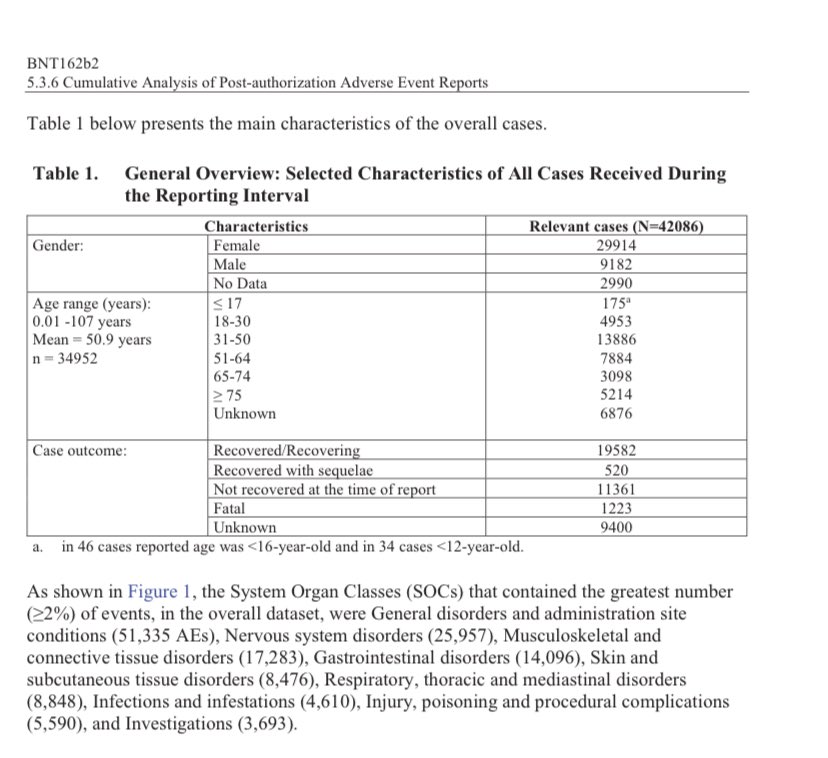
As first reported by Kyle Becker, there were a total of 42,086 case reports for adverse reactions (25,379 medically confirmed, 16,707 non-medically confirmed), spanning 158,893 total events.
More than 25,000 of the events were classified as “Nervous system disorders.”
The original website this document is from and for future releases: Pfizer’s Documents

Since the vaccine has been publicly administered, there have been over 913,000 reports of adverse events in the OpenVAERS global database.
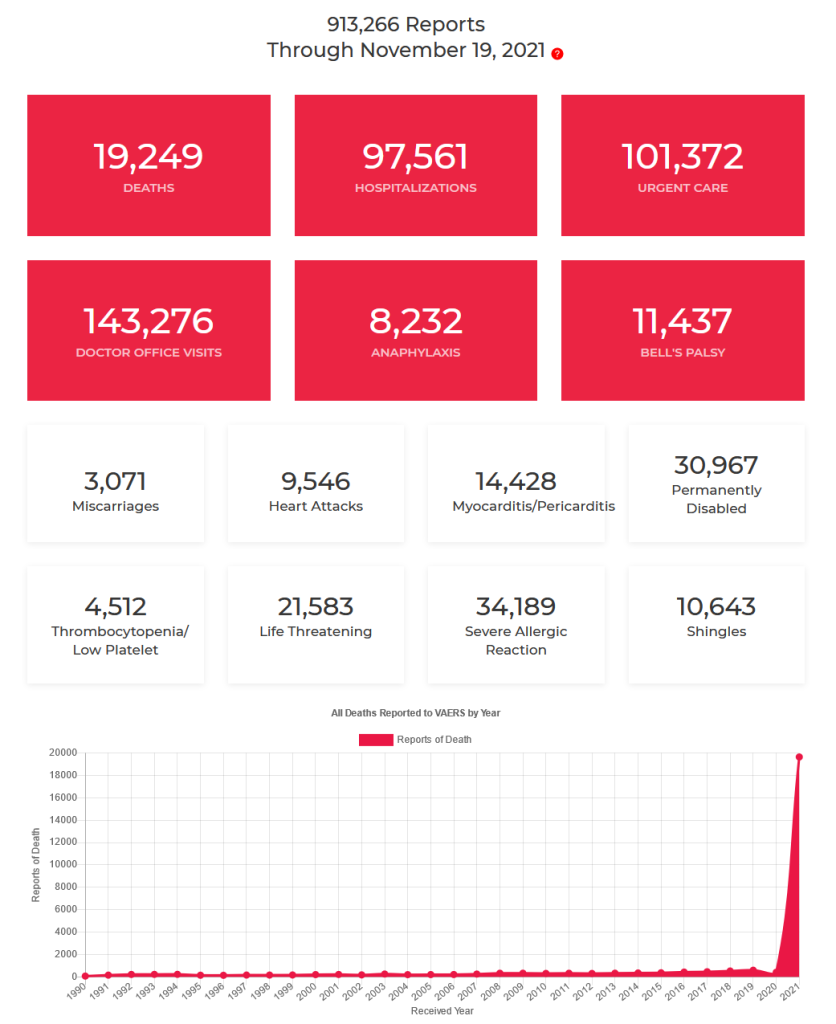
Looking at the amount of AE’s recorded for the 42,000+ is jaw dropping. What’s worse, out of the recorded AE about 1:37 is death. That is insane. The FDA admits these are only recorded AE that are considered above mild. There was so much more that wasn’t released.
The FDA admits these are only recorded AE that are considered above mild. There was so much more that wasn’t released.